
Palladium(II)-Catalyzed Regioselective Hydroesterification of 1,3-Conjugated Enynes with Aryl Formates | Organic Letters
![Palladium-Catalyzed [4 + 3] or [2 + 2 + 3] Annulation via C–H Activation and Subsequent Decarboxylation: Access to Heptagon-Embedded Polycyclic Aromatic Hydrocarbons | Organic Letters Palladium-Catalyzed [4 + 3] or [2 + 2 + 3] Annulation via C–H Activation and Subsequent Decarboxylation: Access to Heptagon-Embedded Polycyclic Aromatic Hydrocarbons | Organic Letters](https://pubs.acs.org/cms/10.1021/acs.orglett.1c00520/asset/images/acs.orglett.1c00520.social.jpeg_v03)
Palladium-Catalyzed [4 + 3] or [2 + 2 + 3] Annulation via C–H Activation and Subsequent Decarboxylation: Access to Heptagon-Embedded Polycyclic Aromatic Hydrocarbons | Organic Letters
![Palladium(II)-Catalyzed Intramolecular [2 + 2 + 2] Annulation of Indolyl 1,3-Diynes: Construction of Azepino-Fused Carbazoles | Organic Letters Palladium(II)-Catalyzed Intramolecular [2 + 2 + 2] Annulation of Indolyl 1,3-Diynes: Construction of Azepino-Fused Carbazoles | Organic Letters](https://pubs.acs.org/cms/10.1021/acs.orglett.3c01186/asset/images/large/ol3c01186_0007.jpeg)
Palladium(II)-Catalyzed Intramolecular [2 + 2 + 2] Annulation of Indolyl 1,3-Diynes: Construction of Azepino-Fused Carbazoles | Organic Letters

A green synthesis of gold–palladium core–shell nanoparticles using orange peel extract through two-step reduction method and its formaldehyde colorimetric sensing performance - ScienceDirect

Green synthesis of Palladium magnetic nanoparticles decorated on carbon nanospheres using Chenopodium and their application as heterogenous catalyst in the Suzuki-Miyaura coupling reaction - ScienceDirect

PDF) Interaction between palladium-doped zerovalent iron nanoparticles and biofilm in granular porous media: Characterization, transport and viability

Direct Access to Palladium(II) Complexes Based on Anionic C,C,C-Phosphonium Ylide Core Pincer Ligand | Inorganic Chemistry

Synthesize palladium nanoparticles from the macroalgae Sargassum fusiforme: An eco-friendly tool in the fight against Plasmodium falciparum? - ScienceDirect
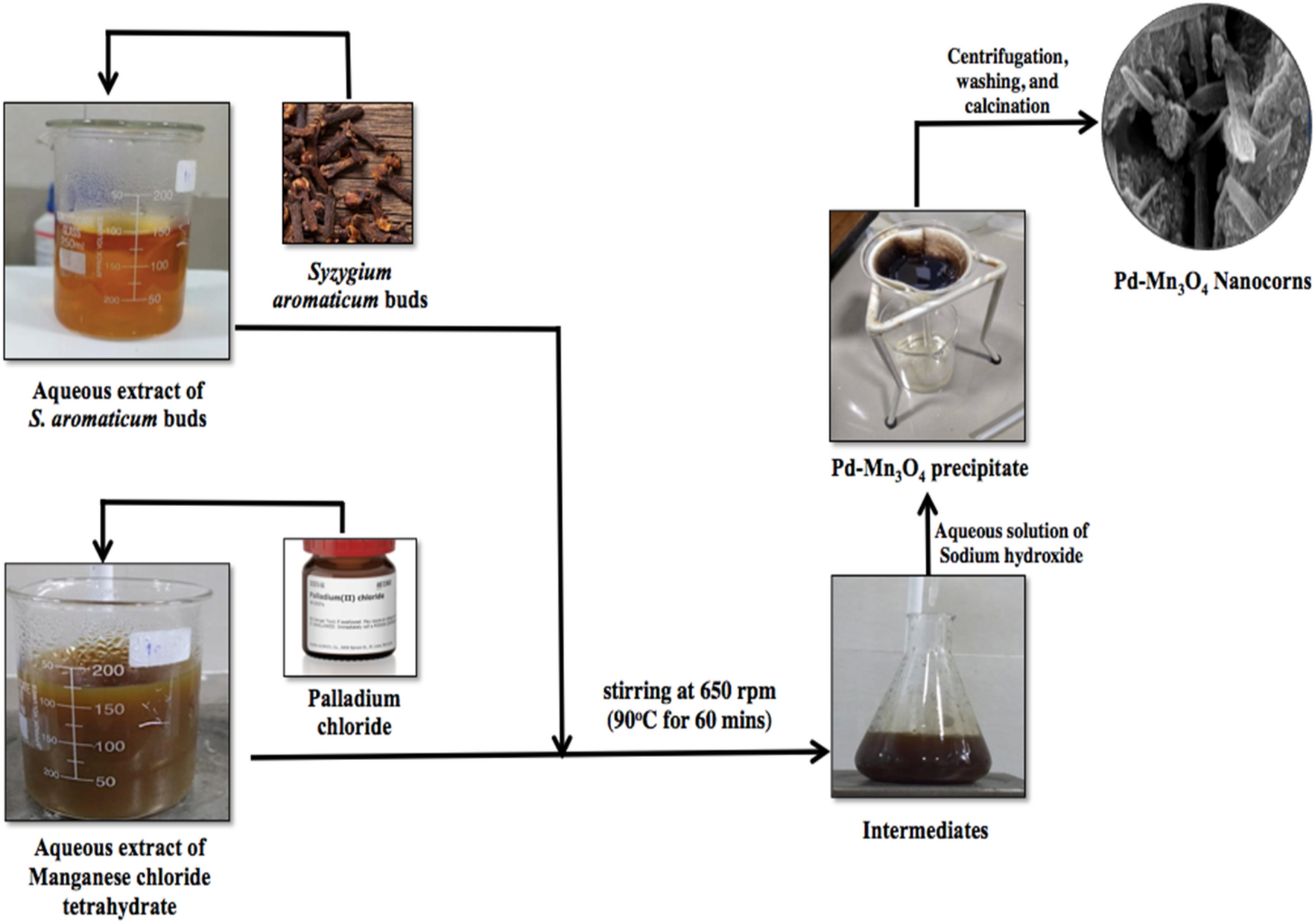
Bioinspired palladium-doped manganese oxide nanocorns: a remarkable antimicrobial agent targeting phyto/animal pathogens | Scientific Reports

Ligand-Centered Triplet Diradical Supported by a Binuclear Palladium(II) Dipyrrindione | Inorganic Chemistry

In-Core N4-Coordination of Palladium(II) in Dinaphthoporphycene: Synthesis, Structure, and Photophysical Studies | Inorganic Chemistry

One-Pot Dual Catalysis of a Photoactive Coordination Polymer and Palladium Acetate for the Highly Efficient Cross-Coupling Reaction via Interfacial Electron Transfer | Inorganic Chemistry

Palladium(II) Pincer Complexes of Functionalized Amides with S-Modified Cysteine and Homocysteine Residues: Cytotoxic Activity and Different Aspects of Their Biological Effect on Living Cells | Inorganic Chemistry
![Palladium-Catalyzed [3 + 2] Annulation of Aryl Halides with 7-Oxa- and 7-Azabenzonorbornadienes via C(sp2 or sp3)–H Activation | Organic Letters Palladium-Catalyzed [3 + 2] Annulation of Aryl Halides with 7-Oxa- and 7-Azabenzonorbornadienes via C(sp2 or sp3)–H Activation | Organic Letters](https://pubs.acs.org/cms/10.1021/acs.orglett.2c03422/asset/images/acs.orglett.2c03422.social.jpeg_v03)










